Effect of Curcumin on HSP Expression in Tilapia Hepatocytes under Hydrogen Peroxide Stress
-
摘要:目的 探究热休克蛋白基因在氧化应激和姜黄素作用下的表达差异,了解姜黄素药效的发挥与热休克蛋白基因表达变化的关系,进一步探讨热休克蛋白对氧化应激条件下鱼类肝细胞中的功能,为鱼类氧化应激的毒理学机制及姜黄素的药理学机制提供理论资料。方法 对24 h姜黄素预处理(保护组)与无姜黄素预处理(对照组)的罗非鱼肝细胞分别进行0、1、2.5 h 的H2O2应激,并对肝细胞5种高分子热休克蛋白基因(HSP70、HSP90α、HSP90β、HSP60、HSP75)和5种小分子热休克蛋白基因(HSP30、HSPB1、 HSPB7、HSPB8、HSPB11)的表达水平进行qPCR检测。结果 与不经H2O2处理(0 h)相比,H2O2作用 1 h能显著提升HSPB1表达量,降低HSP30表达量;H2O2作用2.5 h 能显著提升HSP90a、HSPB1的表达量。姜黄素预处理24 h能显著提升HSP70、HSP90α、HSP30、HSPB1的表达量;姜黄素预处理24 h后再进行H2O2应激1 h时,HSP70、HSP90α、HSP30的表达量显著高于H2O2单独应激(对照组);姜黄素预处理24 h后再进行H2O2应激2.5 h时,HSP90α、HSP30、HSPB1的表达量显著高于H2O2单独应激。其余热休克蛋白在试验过程中无显著差异。结论 H2O2单独应激可引起罗非鱼肝细胞HSP90a、HSP30、HSPB1基因的表达量变化,而姜黄素预处理能提高多种热休克蛋白基因的表达,提高了罗非鱼肝细胞的抗氧化能力,最终抵御H2O2引起的氧化应激并维持细胞活力。Abstract:Objective Expression of heat shock protein (HSP) gene under oxidative stress in the presence of curcumin was studied to understand the toxicological function of the protein and the pharmacological mechanism of curcumin in hepatocytes of Oreochromis nilotica.Methods Tilapia hepatocytes and those pretreated with curcumin for 24 h were subjected to H2O2 stress for 0, 1, or 2.5 h. The expressions of HSP70, HSP90α, HSP90β, HSP60, and HSP75, as well as 5 small molecule HSP genes, i.e., HSP30, HSPB1, HSPB7, HSPB8, and HSPB11, in the cells were determined by qPCR.Results The H2O2 treatment for 1 h significantly increased the expression of HSPB1 and decreased that of HSP30 in the tilapia hepatocytes, while the treatment that lasted for 2.5 h significantly increased the expressions of HSP90a and HSPB1. A pretreatment of curcumin for 24 h on the cells not only significantly elevated the expressions of HSP70, HSP90α, HSP30, and HSPB1 over control but also on those of HSP70, HSP90α, and HSP30 under 1 h H2O2 stress, as well as those on HSP90α, HSP30, and HSPB1 under 2.5 h H2O2 stress. However, no significant differences in other HSPs were observed.Conclusion As a spontaneous response of tilapia hepatocytes to oxidative stress, H2O2 affected significantly the expressions of HSP90a, HSP30, and HSPB1 in the cells. By pretreating the hepatocytes with curcumin, expressions of these genes could be uplifted boosting the cellular antioxidant capacity to better resist the stress and maintain viability.
-
Keywords:
- Tilapia /
- curcumin /
- heat shock proteins /
- oxidative stress
-
0. 引言
【研究意义】热休克蛋白 (Heat shock proteins,HSP) 是一类较为保守的蛋白质超家族。早期人们发现温度上升会使得细胞内的这种蛋白质的表达量升高,于是称其为热休克蛋白[1]。HSP常以分子伴侣的形式参与蛋白质的折叠、转运和降解[2-4]。HSP家族成员众多,根据其分子量的大小可以分为两类,即高分子量HSP(如HSP90、HSP70、HSP60等)和小分子量HSP(sHSP)[5]。sHSP又包含有组织特异性表达的HSPB2、HSPB3、HSPB4、HSPB7等和无组织特异性表达的HSPB1、HSPB5、HSPB6、HSPB8等[5-6]。鱼类作为一种重要的水生动物,其对水环境中的温度、盐度、氧气等环境因子高度依赖。面对复杂环境,热休克蛋白的调节对鱼类适应环境有着特殊的意义。越来越多的研究表明热休克蛋白能诱导鱼类细胞响应各种环境因子胁迫,如高温[7]、低氧[8]、声波[9]、污染物暴露[10-12]、病原体感染[13]等。因此,探究热休克蛋白在鱼类对抗应激方面的作用机制对于鱼类的毒理学研究和药物研发有着现实意义。【前人研究进展】外界环境的不利因素会引起养殖鱼类的氧化应激[14]。氧化应激产生的活性氧(Reactive oxygen species,ROS)作用于蛋白质会导致氨基酸碳链断裂、二硫键形成以及其他的氧化修饰,从而改变蛋白质的高级结构,最终导致蛋白质失活。热休克蛋白常参与蛋白质的折叠与对错误折叠蛋白质的降解等生物学过程,因此在氧化应激过程中构象异常的蛋白质能否被热休克蛋白重新折叠或降解是维持细胞存活的必要条件之一[15]。研究表明氧化应激能改变鱼类热休克蛋白的基因表达,例如,铜和镉作用下鲤(Cyprinus carpio)脑与肝脏中HSP60、HSP70和HSP90的基因表达和蛋白质含量出现不同程度的升高[11]。另一方面,上调热休克蛋白的基因表达有益于细胞抵抗氧化应激。Franco等[16]发现外源热休克蛋白能保护人淋巴瘤细胞免于H2O2引起的死亡。过表达HSP70能提高心肌细胞内超氧化物歧化酶的活性,降低ROS引起的损伤[17]。过表达HSP70能通过抑制细胞凋亡信号通路,减少大鼠肝细胞因H2O2引起的氧化损伤[18]。在骨肉瘤细胞(Saos-2)的研究中发现,高表达HSP75有助于抑制H2O2造成的DNA损伤[19]。在鱼类的研究中,热休克蛋白基因(HSP70和HSP90)表达的上升反映了细胞对氨氮造成的氧化应激的防御性反应[20]。此外,鲤鱼在重金属暴露下肝脏内的HSP70和HSP90表达呈现上升的趋势[21]。姜黄素(Curcumin)是植物姜黄中的有效成分,是一种具有抗氧化、抗炎症、抗癌功能的多元酚,能通过NF-κB、Nrf2等转录因子调节相关的信号通路起作用,在养殖中常用于鱼类的保肝护肝[22],但其机制还不清楚。【本研究切入点】笔者在先前的研究中发现姜黄素预处理能保护H2O2应激下罗非鱼肝细胞的活力,并通过上调Nrf2信号通路提高抗氧化酶活力和血红素加氧酶1(Heme Oxygenase-1,HO-1)的基因的表达量[22],而HO-1也是热休克蛋白的一种(HSP32)。此外其他研究也表明姜黄素对热休克蛋白也有调节功能[23]。本文在前期研究的基础上进一步探究在氧化应激条件下姜黄素对罗非鱼热休克蛋白基因表达的影响,分析姜黄素的应激保护水平。【拟解决的关键问题】在罗非鱼体外肝细胞氧化应激模型下,采用姜黄素进行预处理,研究姜黄素预处理保护对罗非鱼肝细胞氧化应激过程中热休克蛋白家族基因表达的影响,为鱼类氧化应激的毒理学机制及姜黄素的药理学机制提供理论资料。
1. 材料与方法
1.1 试验材料
成年罗非鱼由福建省淡水水产研究所榕桥试验基地提供。罗非鱼暂养于100 L玻璃养殖缸内2周,以适应实验室养殖条件。期间保持养殖水体持续曝气并维持水温于28 °C,每日投喂饲料2次。
姜黄素购于Sigma-Aldrich公司。将姜黄素预溶解于二甲基亚砜(DMSO)中,再用DMEM/F12细胞培养液溶解姜黄素至工作浓度(40 μmol∙L−1),同时确保DMSO在细胞培养液中的终浓度小于0.1%。
1.2 罗非鱼原代肝细胞培养
罗非鱼原代肝细胞培养方法见先前发表的论文[22],具体方法如下:挑选体色正常、活力好的罗非鱼,经MS-222(100 mg∙L−1)麻醉后用注射器插入心脏放血。75%医用酒精消毒体表后迅速剖开鱼腹取出肝脏并置于无菌培养皿中。将肝脏组织切碎成约1 mm3的组织块,并去除脂肪、血管等无关的组织。Hank’s缓冲液反复清洗组织块至血色减退。0.25%胰蛋白酶溶液消化肝脏组织(28 ℃),共消化3次,每次15 min,并在每个消化终点收集肝细胞。细胞悬液经75 μm不锈钢筛过滤后得到单细胞悬液。将此细胞悬液在50×g离心力下低速离心3 min,收集细胞沉淀,重复2次。DMEM/F12培养液(含100 IU∙mL−1青霉素、100 mg∙mL−1链霉素和10%胎牛血清)重新悬浮细胞,并调整密度为1×106 个∙mL−1,接种于6孔板中,放入CO2培养箱培养。培养箱温度设定为28 ℃,CO2浓度为5%。
1.3 试验处理
试验分为对照组和保护组。对照组细胞在试验前24 h正常更换培养液,保护组则用含40 μmol∙L−1姜黄素的培养液预处理24 h。24 h过后2组分别采集样品并标记为第一个时间点(0 h),随后分别用5 mmol∙L−1 H2O2处理1 h和2.5 h,并在作用时间终点采样。每个样品约为2×106个细胞。
1.4 RNA提取及热休克蛋白家族各基因qPCR检测
罗非鱼肝细胞经药物处理后,去除培养液。每孔加入1 mL 磷酸缓冲液清洗并去除细胞碎屑,再加入1 mL Trizol试剂(Invitrogen,USA)并反复吹打至细胞全部脱落。按说明书步骤提取总RNA。总RNA经电泳检测后,采用NanoDrop检测样品的OD值与核酸浓度。采用反转录试剂盒(Yeasen,中国上海)制备各样品cDNA模板。
采用LightCycler 480 Roche实时热循环仪的384孔模块对各组细胞cDNA模板进行qPCR检测,反应体系为10 μL,包含上下游引物(引物序列参见表1)(10 μmol∙L−1)各0.25 μL,5 μL SYBR Green Master Mix (Promega,USA)和4.5 μL cDNA模板。用于PCR的循环条件如下:在95 °C下1 min,95 °C 15 s(40次循环),60 °C孵育1 min。数据结果采用2−ΔΔCt值方法计算基因相对表达水平[2]。
表 1 qPCR检测所用的各基因引物Table 1. Primers used for qPCR基因
Gene长度
Length/bp引物序列(5′–3′)
Sequence (5′–3′)HSP70 107 HSP70F: CATCGCCTACGGTCTGGACAA HSP70R: TGCCGTCTTCAATGGTCAGGAT HSP90α 128 HSP90αF: ATTGCTCAGCTGATGTCCCT HSP90αR: GTGGGATCCGTCAAGCTTTC HSP90β 117 HSP90βF: AGAACCTCAAGCTGGGTGTG HSP90βR: TCACGTACTCCGAGAGGGAG HSP60 115 HSP60F: GCGGCTAGCAGTAAGCCTTT HSP60R: CGTAGGCTCGTGTGAGGTG HSP75 103 HSP75F: GTTGGGCCCACCATTCATCC HSP75R: TTCTCAACCATCCACGCCAT HSP30 111 HSP30F: GACTCTGGACCTCCTCCACT HSP30R: TGAAACACTCTTCAGGTGTCTGT HSPB1 106 HSPB1F: CTTTGACCAGACCTTCGGCA HSPB1R: TAGTTCTGGGGCCAGGATGG HSPB7 119 HSPB7F: ACTTTCACCCACAAGTGCCA HSPB7R: AGCTCATGCTTGGCTGGATT HSPB8 104 HSPB8F: TGGAAAGTCTGCGTGAACGTC HSPB8R: TCCTGTTTCTCTTCGTGCTTTC HSPB11 100 HSPB11F: CAGCTTGTTTGGCGATGACC HSPB11R: CCGGTTGAAGAAGTCTCGCT β-Actin 129 β-Actin-F: TGCGGGATATCATTTGCCTGA β-Actin-R: GAATCCGGCCTTGCACATAC 1.5 数据处理
使用IBM SPSS Statistics 19软件对数据进行统计学分析,t检验确定同一时间不同处理的2个样本之间是否存在显著差异(P < 0.05)。采用单因素方差分析和Tukey’ s HSD方法分析同一处理组不同时间点之间的显著差异(P < 0.05)。
2. 结果与分析
2.1 姜黄素预处理下高分子量HSP的基因表达差异
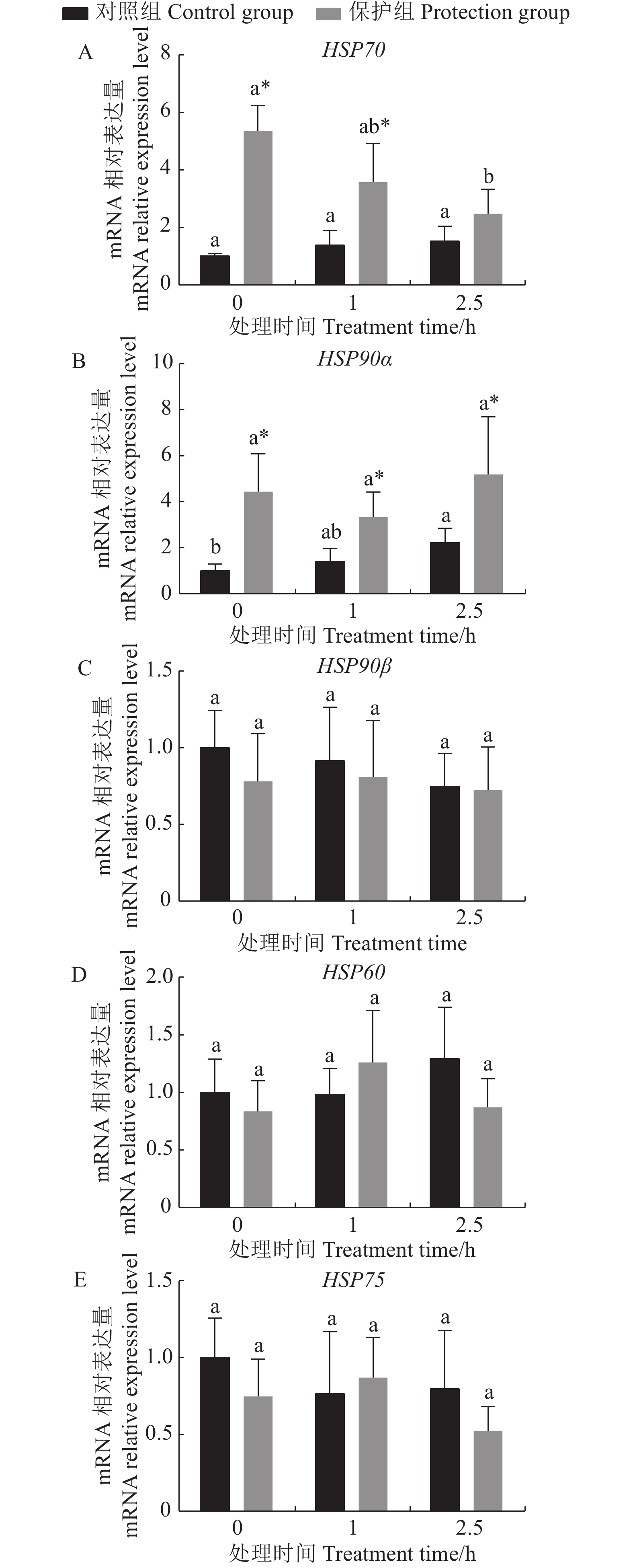
不同处理对高分子量热休克蛋白HSP70、HSP90α、HSP90β、HSP60、HSP75的基因相对表达量的影响如图1所示。各处理HSP90β、HSP75、HSP60基因表达量在氧化应激作用下变化趋势不明显,且保护组与对照组间并无显著性差异(P>0.05)。保护组HSP70在H2O2处理下呈逐渐下降的趋势,对照组呈逐渐升高的趋势;保护组HSP70在H2O2处理0、1 h的表达显著高于对照组(P<0.05),而处理2.5 h与对照组无显著差异(P>0.05)(图1-A)。说明姜黄素提高了罗非鱼肝细胞中HSP70的表达量,并在H2O2应激1 h内维持HSP70的高表达,而单纯H2O2应激不会引起罗非鱼肝细胞HSP70的表达显著变化。
![]() 图 1 姜黄素预处理对H2O2胁迫下罗非鱼肝细胞高分子HSP基因表达的影响*表示同一处理时间保护组与对照组间差异显著(P < 0.05),不同字母小写字母表示同一处理组中不同时间点之间相比差异显著(P < 0.05)。图2同。Figure 1. Effect of curcumin pretreatment on HSP expressions of tilapia hepatocytes under H2O2 stress* indicates significant difference from control at relevant time point at P<0.05; data with different lowercase letters indicate significant differences at different time points in same group at P<0.05. Same for Fig. 2.
图 1 姜黄素预处理对H2O2胁迫下罗非鱼肝细胞高分子HSP基因表达的影响*表示同一处理时间保护组与对照组间差异显著(P < 0.05),不同字母小写字母表示同一处理组中不同时间点之间相比差异显著(P < 0.05)。图2同。Figure 1. Effect of curcumin pretreatment on HSP expressions of tilapia hepatocytes under H2O2 stress* indicates significant difference from control at relevant time point at P<0.05; data with different lowercase letters indicate significant differences at different time points in same group at P<0.05. Same for Fig. 2.氧化应激作用下,保护组HSP90α表达量呈先降低后升高的趋势,对照组则呈现缓慢上升趋势,2.5 h的表达量显著高于0 h;各个时间点保护组HSP90α均显著高于对照组(P<0.05)(图1-B)。说明姜黄素提升了肝细胞中HSP 90α的表达量。此外对照组H2O2应激后2.5 h的HSP90α表达水平显著高于应激前(0 h)(P<0.05),表明H2O2应激2.5 h也能显著提升肝细胞HSP90α的表达水平,但表达水平低于姜黄素预处理后再进行H2O2应激2.5 h的肝细胞(图1-B)。
2.2 姜黄素预处理下小分子HSP的基因表达差异
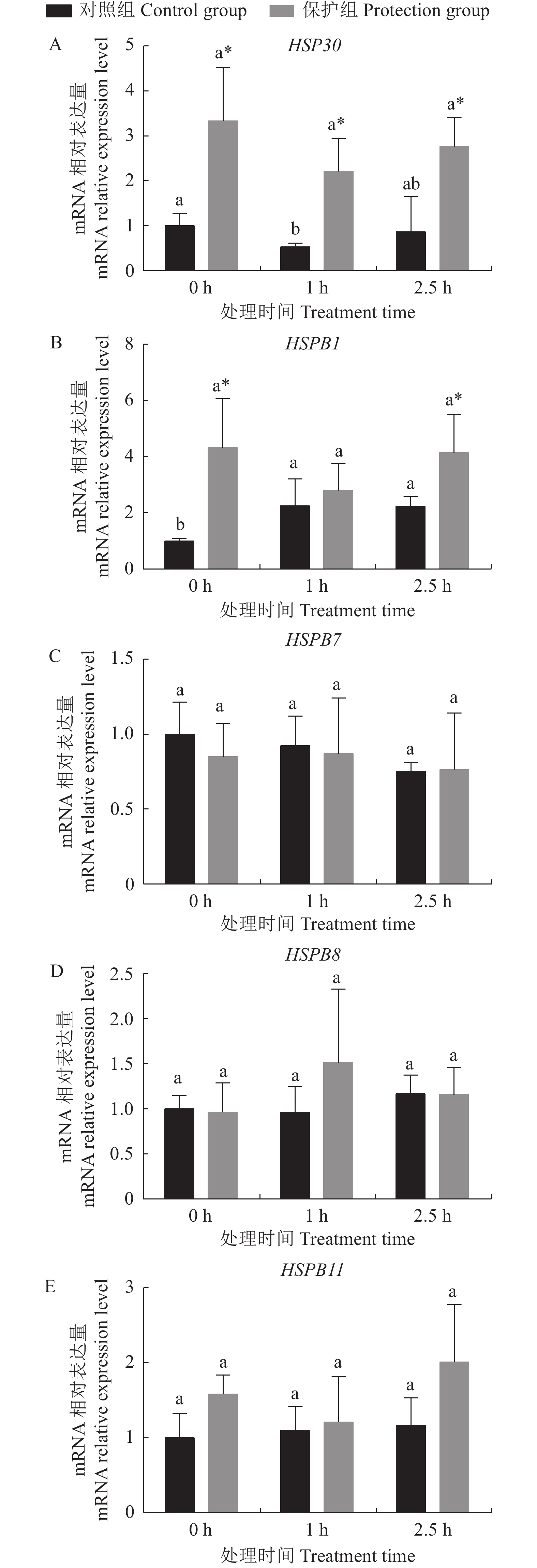
不同处理对高分子量热休克蛋白HSP30、HSPB1、HSPB7、HSPB8、HSPB11的基因相对表达量的影响如图2所示。各处理HSPB7、HSPB8、HSPB11在氧化应激作用下变化趋势不明显,且保护组与对照组间无显著性差异(图2-C、D、E)。保护组HSP30的表达量在氧化应激的作用下变化不显著,但显著高于对照组(图2-A)(P<0.05),说明承受氧化应激前后阶段姜黄素都能提升细胞HSP30的表达量;对照组HSP30的基因表达呈现先下降后上升趋势,H2O2应激后1 h HSP30显著低于应激前(0 h)(P<0.05),而应激2.5 h后HSP30的表达量又恢复到应激前水平(图2-A)。
对照组HSPB1基因表达量在H2O2应激后显著上升,1 h 和2.5 h 的HSPB1表达量与0 h相比显著升高 (P<0.05),即H2O2应激导致了细胞HSPB1表达量的上升。保护组HSPB1基因表达量在氧化应激各个时间点差异不显著(P>0.05)。保护组HSPB1在应激前和应激后2.5 h时显著高于对照组(P<0.05),说明姜黄素能提升肝细胞在H2O2应激时的HSPB1的表达(图2-B)。
3. 讨论
3.1 H2O2诱导的氧化应激对罗非鱼肝细胞HSP基因表达的影响
HSP90蛋白家族是一类进化上高度保守的分子家族,参与许多细胞生物学过程[24]。研究表明,在氧化应激条件下,鱼类细胞中HSP90的基因表达发生了变化。在鲤鱼肝细胞中,H2O2诱导的氧化应激显著改变了超氧化物歧化酶和过氧化氢酶活性,并增强了HSP90的表达[25]。水体中的H2O2暴露后使鲤鱼不同组织中碱性磷酸酶、酸性磷酸酶、溶菌酶的活性和HSP90基因的表达产生显著差异[26]。本研究中急性H2O2应激(2.5 h)可提高HSP90α的表达,但对HSP90β无显著影响。这可能是由于当细胞受到应激时,HSP90α往往倾向于受应激诱导而呈现基因表达的变化,而HSP90β表现为组成性表达,不易随应激而变化[27-28]。 另一项研究得到了与本研究相似的结果:高氨诱导的氧化应激可显著提高黄鳝(Monopterus cuchia)HSP90α mRNA基因及其翻译蛋白质的水平;而HSP90β的表达水平无明显变化[29]。HSP75是HSP90家族的一员,定位于线粒体基质中[24]。HSP75可以维持线粒体的完整性,防止过多ROS引起的细胞死亡[24]。在本研究中,HSP75在各组间无显著差异表达。鉴于目前对鱼类HSP75的研究相对较少,未来应进一步研究鱼类HSP75的抗氧化应激相关机制。
HSPB1被认为能降低细胞内ROS水平、维持谷胱甘肽(GSH)水平并在稳定线粒体膜电位中发挥重要作用[30]。在大鼠(Rattus norvegicus)心肌细胞中,HSPB1能改善包括谷胱甘肽还原酶(Glutathione reductase,GR)、谷胱甘肽过氧化物酶(Glutathion peroxidase,GPx)和硫氧还蛋白(Thioredoxin,Trx)的抗氧化系统[30-31]。目前,在硬骨鱼的多个物种中已经鉴定出HSPB1,有学者观察到鱼类在病毒感染过程中HSPB1的表达增加的现象[32-33]。目前鱼类HSPB1的抗氧化机制尚不清楚。在本研究中,H2O2显著增加了HSPB1的表达,而HSPB1的高表达有助于细胞激活抗氧化系统。因此可以推测, HSPB1表达的显著增加是罗非鱼肝细胞对抗氧化应激的自发防御。
3.2 姜黄素对罗非鱼肝细胞的保护作用
直接或间接增加细胞内热休克蛋白的含量有助于提升细胞的抗氧化能力[34-36]。因此,许多药物通过诱导热休克蛋白的表达来发挥药效,热休克蛋白也常作为相关药物作用的靶标[24]。研究表明姜黄素对热休克蛋白的表达有促进作用,姜黄素能上调低温应激下的胰岛组织中的HSP70并保护胰岛组织的完整性[37]。另一项研究表明姜黄素有助于提升人肺癌组织中HSP70的表达水平[38]。饲喂姜黄素和白藜芦醇(resveratrol)能有效提高大鼠睾丸的HSP60、HSP70与HSP90表达量,并减轻邻苯二甲酸二乙基己酯(Diethylhexyl phthalate,DEHP)引起的氧化应激与睾丸损伤[39]。这些研究说明姜黄素对一些热休克蛋白的表达有促进作用,这与本研究结果是一致的。本试验中姜黄素预处理能提升HSP70、HSP90α、HSP30、HSPB1的表达量,而这种提升在有H2O2应激的情况下仍然维持较高水平并显著高于H2O2单独作用,表明姜黄素可能通过提升相关热休克蛋白的表达量来提升鱼类肝细胞的抗氧化能力,最终保护肝细胞活力。笔者在前期研究发现姜黄素能显著提高罗非鱼肝细胞氧化应激模型下的细胞活力,姜黄素的这种保护作用与其对肝细胞氧化应激通路Nrf2的调控密切相关[22],而本研究结果显示姜黄素的这种保护作用还和其对HSP的调控有关。至于姜黄素提升细胞热休克蛋白表达量的相关机制还需进一步研究。近年研究表明,姜黄素通过提高HSP70的转录因子——热休克因子1(HSF1)的磷酸化水平,有效提高人肠上皮细胞HSP70水平,从而增强肠上皮的完整性[23]。在医学研究中,姜黄素被认为是HSF1和Nrf2的双重激活剂[40]。这为研究姜黄素与HSPs的调控机制提供了新的思路,今后可从热休克蛋白的上游转录因子入手进行更深入的研究。
-
图 1 姜黄素预处理对H2O2胁迫下罗非鱼肝细胞高分子HSP基因表达的影响
*表示同一处理时间保护组与对照组间差异显著(P < 0.05),不同字母小写字母表示同一处理组中不同时间点之间相比差异显著(P < 0.05)。图2同。
Figure 1. Effect of curcumin pretreatment on HSP expressions of tilapia hepatocytes under H2O2 stress
* indicates significant difference from control at relevant time point at P<0.05; data with different lowercase letters indicate significant differences at different time points in same group at P<0.05. Same for Fig. 2.
表 1 qPCR检测所用的各基因引物
Table 1 Primers used for qPCR
基因
Gene长度
Length/bp引物序列(5′–3′)
Sequence (5′–3′)HSP70 107 HSP70F: CATCGCCTACGGTCTGGACAA HSP70R: TGCCGTCTTCAATGGTCAGGAT HSP90α 128 HSP90αF: ATTGCTCAGCTGATGTCCCT HSP90αR: GTGGGATCCGTCAAGCTTTC HSP90β 117 HSP90βF: AGAACCTCAAGCTGGGTGTG HSP90βR: TCACGTACTCCGAGAGGGAG HSP60 115 HSP60F: GCGGCTAGCAGTAAGCCTTT HSP60R: CGTAGGCTCGTGTGAGGTG HSP75 103 HSP75F: GTTGGGCCCACCATTCATCC HSP75R: TTCTCAACCATCCACGCCAT HSP30 111 HSP30F: GACTCTGGACCTCCTCCACT HSP30R: TGAAACACTCTTCAGGTGTCTGT HSPB1 106 HSPB1F: CTTTGACCAGACCTTCGGCA HSPB1R: TAGTTCTGGGGCCAGGATGG HSPB7 119 HSPB7F: ACTTTCACCCACAAGTGCCA HSPB7R: AGCTCATGCTTGGCTGGATT HSPB8 104 HSPB8F: TGGAAAGTCTGCGTGAACGTC HSPB8R: TCCTGTTTCTCTTCGTGCTTTC HSPB11 100 HSPB11F: CAGCTTGTTTGGCGATGACC HSPB11R: CCGGTTGAAGAAGTCTCGCT β-Actin 129 β-Actin-F: TGCGGGATATCATTTGCCTGA β-Actin-R: GAATCCGGCCTTGCACATAC -
[1] 郭文亮, 徐元庆, 金晓, 等. 热休克蛋白在冷应激引起的炎症反应和氧化应激中的调节作用 [J]. 畜牧兽医学报, 2022, 53(6):1668−1677. DOI: 10.11843/j.issn.0366-6964.2022.06.002 GUO W L, XU Y Q, JIN X, et al. Moderating role of heat shock protein under inflammatory response and oxidative stress caused by cold stress [J]. Acta Veterinaria et Zootechnica Sinica, 2022, 53(6): 1668−1677.(in Chinese) DOI: 10.11843/j.issn.0366-6964.2022.06.002
[2] ZHANG X, LI Y T, SUN Y L, et al. Regulatory effect of heat shock transcription factor-1 gene on heat shock proteins and its transcriptional regulation analysis in small abalone Haliotis diversicolor [J]. BMC Molecular and Cell Biology, 2020, 21(1): 83. DOI: 10.1186/s12860-020-00323-9
[3] LI Y T, ZHANG T, ZHANG X, et al. Heat shock cognate 70 gene in Haliotis diversicolor: Responses to pathogen infection and environmental stresses and its transcriptional regulation analysis [J]. Cell Stress and Chaperones, 2018, 23(3): 335−346. DOI: 10.1007/s12192-017-0842-5
[4] FANG Z Q, SUN Y L, ZHANG X, et al. Responses of HSP70 gene to Vibrio parahaemolyticus infection and thermal stress and its transcriptional regulation analysis in Haliotis diversicolor [J]. Molecules, 2019, 24(1): 162. DOI: 10.3390/molecules24010162
[5] 李艳, 张国强, 邵东燕, 等. 小分子热休克蛋白的结构与功能 [J]. 生命科学, 2017, 29(1):55−61. LI Y, ZHANG G Q, SHAO D Y, et al. The structure and function of the small heat shock proteins [J]. Chinese Bulletin of Life Sciences, 2017, 29(1): 55−61.(in Chinese)
[6] 李婷婷, 孙继红, 韩森荣, 等. 水产动物小分子热休克蛋白家族研究进展 [J]. 水产学杂志, 2022, 35(4):101−109. DOI: 10.3969/j.issn.1005-3832.2022.04.015 LI T T, SUN J H, HAN S R, et al. A review: Research progress on small molecule heat shock protein family in animals in aquaculture [J]. Chinese Journal of Fisheries, 2022, 35(4): 101−109.(in Chinese) DOI: 10.3969/j.issn.1005-3832.2022.04.015
[7] 张晨光, 丁炜东, 曹哲明, 等. 急性高温胁迫对翘嘴鳜幼鱼抗氧化酶和消化酶活性及热休克蛋白基因表达的影响 [J]. 南方农业学报, 2021, 52(3):815−826. DOI: 10.3969/j.issn.2095-1191.2021.03.030 ZHANG C G, DING W D, CAO Z M, et al. Effects of acute high temperature stress on antioxidant enzymes activity, digestive enzymes activity and gene expression of heat shock proteins in mandarin fish(Siniperca chuatsi) [J]. Journal of Southern Agriculture, 2021, 52(3): 815−826.(in Chinese) DOI: 10.3969/j.issn.2095-1191.2021.03.030
[8] 狄治朝, 周涛, 许强华. 低氧胁迫与常氧条件下斑马鱼鳃中热休克蛋白基因家族的表达差异比较 [J]. 大连海洋大学学报, 2018, 33(6):690−695. DI Z C, ZHOU T, XU Q H. Expression comparisons of heat shock proteins gene in gills of zebrafish exposed to both normoxia and hypoxia [J]. Journal of Dalian Ocean University, 2018, 33(6): 690−695.(in Chinese)
[9] 林海霞. 声胁迫对斜带石斑鱼生理生化指标和Hsp70表达的影响[D]. 湛江: 广东海洋大学, 2019. LIN H X. Effects of acoustic stress on physiological and biochemical indexes and Hsp70 expression of Epinephelus coioides[D]. Zhanjiang: Guangdong Ocean University, 2019. (in Chinese)
[10] 贾利兴. 壬基酚暴露对斑马鱼热休克蛋白表达量的影响[D]. 广州: 暨南大学, 2016. JIA L X. Effects of exposure to nonylphenol on expression of heat shock protein of zebrafish[D]. Guangzhou: Jinan University, 2016. (in Chinese)
[11] 姜旭阳. 铜和镉单一及联合亚急性暴露对鲤幼鱼热休克蛋白的影响[D]. 哈尔滨: 东北农业大学, 2015. JIANG X Y. Effects of single and joint subacute exposure of copper and cadmium on heat shock proteins in common carp(Cyprinus carpio)[D]. Harbin: Northeast Agricultural University, 2015. (in Chinese)
[12] LI Z, ALI SHAH S W, ZHOU Q, et al. The contributions of miR-25-3p, oxidative stress, and heat shock protein in a complex mechanism of autophagy caused by pollutant cadmium in common carp (Cyprinus carpio L. ) hepatopancreas [J]. Environmental Pollution, 2021, 287: 117554. DOI: 10.1016/j.envpol.2021.117554
[13] CHENG J, LI H H, HUANG Z Y, et al. Expression analysis of the heat shock protein genes and cellular reaction in dojo loach (Misgurnus anguillicaudatus) under the different pathogenic invasion [J]. Fish & Shellfish Immunology, 2019, 95: 506−513.
[14] 周俊豪, 刘念, 杨映. 硬骨鱼类Keap1-Nrf2/ARE信号通路研究进展 [J]. 水产学杂志, 2022, 35(4):117−124. DOI: 10.3969/j.issn.1005-3832.2022.04.018 ZHOU J H, LIU N, YANG Y. Keap1-Nrf2/ARE signaling pathway and its antioxidant regulation in teleost fish: A review [J]. Chinese Journal of Fisheries, 2022, 35(4): 117−124.(in Chinese) DOI: 10.3969/j.issn.1005-3832.2022.04.018
[15] 柯湘宇. 氧化应激中热休克蛋白90对MRN复合物相关的DNA双链断裂损伤修复通路的调控机制研究[D]. 广州: 南方医科大学, 2017. KE X Y. The role of Hsp90 in MRE11-RAD50-NBS1 complex related DNA double strands break repair signaling pathway in oxidative stress[D]. Guangzhou: Southern Medical University, 2017. (in Chinese)
[16] FRANCO L, TERRINCA J, RODRÍGUEZ A B, et al. Extracellular heat shock proteins protect U937 cells from H2O2-induced apoptotic cell death [J]. Molecular and Cellular Biochemistry, 2016, 412(1): 19−26.
[17] SABIRZHANOV B, STOICA B A, HANSCOM M, et al. Over-expression of HSP70 attenuates caspase-dependent and caspase-independent pathways and inhibits neuronal apoptosis [J]. Journal of Neurochemistry, 2012, 123(4): 542−554. DOI: 10.1111/j.1471-4159.2012.07927.x
[18] 王慧. 腺病毒介导的hsp70过表达对H2O2诱导BRL细胞凋亡的保护作用研究[D]. 大庆: 黑龙江八一农垦大学, 2014. WANG H. Study on the protective effect of adenovirus-mediated transfer of hsp70 in BRL cells against apoptosis by hydrogen peroxide[D]. Daqing: Heilongjiang Bayi Agricultural University, 2014. (in Chinese)
[19] MONTESANO GESUALDI N, CHIRICO G, PIROZZI G, et al. Tumor necrosis factor-associated protein 1 (TRAP-1) protects cells from oxidative stress and apoptosis [J]. Stress, 2007, 10(4): 342−350. DOI: 10.1080/10253890701314863
[20] CHENG C H, YANG F F, LING R Z, et al. Effects of ammonia exposure on apoptosis, oxidative stress and immune response in pufferfish (Takifugu obscurus) [J]. Aquatic Toxicology, 2015, 164: 61−71. DOI: 10.1016/j.aquatox.2015.04.004
[21] MONIRUZZAMAN M, MIDDAY P, DHARA A, et al. Change in redox state and heat shock protein expression in an Indian major carp Cirrhinus cirrhosus exposed to zinc and lead [J]. The Journal of Toxicological Sciences, 2017, 42(6): 731−740. DOI: 10.2131/jts.42.731
[22] LI L M, ZHANG Z P, HUANG Y F. Integrative transcriptome analysis and discovery of signaling pathways involved in the protective effects of curcumin against oxidative stress in tilapia hepatocytes [J]. Aquatic Toxicology, 2020, 224: 105516. DOI: 10.1016/j.aquatox.2020.105516
[23] GUO M Z, XU W X, YAMAMOTO Y, et al. Curcumin increases heat shock protein 70 expression via different signaling pathways in intestinal epithelial cells [J]. Archives of Biochemistry and Biophysics, 2021, 707: 108938. DOI: 10.1016/j.abb.2021.108938
[24] HOTER A, EL-SABBAN M E, NAIM H Y. The HSP90 family: Structure, regulation, function, and implications in health and disease [J]. International Journal of Molecular Sciences, 2018, 19(9): 2560. DOI: 10.3390/ijms19092560
[25] MONIRUZZAMAN M, GHOSAL I, DAS D, et al. Melatonin ameliorates H2O2-induced oxidative stress through modulation of Erk/Akt/NFkB pathway [J]. Biological Research, 2018, 51(1): 17. DOI: 10.1186/s40659-018-0168-5
[26] JIA R, DU J L, CAO L P, et al. Immune, inflammatory, autophagic and DNA damage responses to long-term H2O2 exposure in different tissues of common carp (Cyprinus carpio) [J]. The Science of the Total Environment, 2021, 757: 143831. DOI: 10.1016/j.scitotenv.2020.143831
[27] BUGG W S, YOON G R, BRANDT C, et al. The effects of population and thermal acclimation on the growth, condition and cold responsive mRNA expression of age-0 lake sturgeon (Acipenser fulvescens) [J]. Journal of Fish Biology, 2021, 99(6): 1912−1927. DOI: 10.1111/jfb.14897
[28] 段丹萍. Hsp90α与过氧化氢诱导的人肝癌细胞和人正常肝细胞氧化应激损伤的相关性探讨[D]. 广州: 南方医科大学, 2013. DUAN D P. Hsp90α is involved in stress response of HepG2 liver carcinoma cells and L02 hepatocytes to hydrogen peroxide induced oxidative stress[D]. Guangzhou: Southern Medical University, 2013. (in Chinese)
[29] HANGZO H, BANERJEE B, SAHA S, et al. Ammonia stress under high environmental ammonia induces Hsp70 and Hsp90 in the mud eel, Monopterus cuchia [J]. Fish Physiology and Biochemistry, 2017, 43(1): 77−88. DOI: 10.1007/s10695-016-0269-4
[30] LIU X H, XIAO W, JIANG Y, et al. Bmal1 regulates the redox rhythm of HSPB1, and homooxidized HSPB1 attenuates the oxidative stress injury of cardiomyocytes [J]. Oxidative Medicine and Cellular Longevity, 2021, 2021: 5542815.
[31] TERRA L F, WAILEMANN R A M, DOS SANTOS A F, et al. Heat shock protein B1 is a key mediator of prolactin-induced beta-cell cytoprotection against oxidative stress [J]. Free Radical Biology and Medicine, 2019, 134: 394−405. DOI: 10.1016/j.freeradbiomed.2019.01.023
[32] LE Y, JIA P, JIN Y L, et al. The antiviral role of heat shock protein 27 against red spotted grouper nervous necrosis virus infection in sea perch [J]. Fish & Shellfish Immunology, 2017, 70: 185−194.
[33] LI P H, CAI Y J, ZHU X L, et al. Epinephelus coioides Hsp27 negatively regulates innate immune response and apoptosis induced by Singapore grouper iridovirus (SGIV) infection [J]. Fish & Shellfish Immunology, 2022, 120: 470−480.
[34] KIM W, KWON H J, JUNG H Y, et al. Tat-HSP70 protects neurons from oxidative damage in the NSC34 cells and ischemic damage in the ventral horn of rabbit spinal cord [J]. Neurochemistry International, 2019, 129: 104477. DOI: 10.1016/j.neuint.2019.104477
[35] KIM W K, KIM W H, KWEON O K, et al. Heat-Shock Proteins can potentiate the therapeutic ability of cryopreserved mesenchymal stem cells for the treatment of acute spinal cord injury in dogs [J]. Stem Cell Reviews and Reports, 2022, 18(4): 1461−1477.
[36] CHEN Z Y, SHEN X L, SHEN F Y, et al. TAK1 activates AMPK-dependent cell death pathway in hydrogen peroxide-treated cardiomyocytes, inhibited by heat shock protein-70 [J]. Molecular and Cellular Biochemistry, 2013, 377(1): 35−44.
[37] KANITKAR M, BHONDE R R. Curcumin treatment enhances islet recovery by induction of heat shock response proteins, Hsp70 and heme oxygenase-1, during cryopreservation [J]. Life Sciences, 2008, 82(3/4): 182−189.
[38] CHEN H W, YU S L, CHEN J J W, et al. Anti-invasive gene expression profile of curcumin in lung adenocarcinoma based on a high throughput microarray analysis [J]. Molecular Pharmacology, 2004, 65(1): 99−110. DOI: 10.1124/mol.65.1.99
[39] ABD EL-FATTAH A A, FAHIM A T, ABDEL HAMID SADIK N, et al. Resveratrol and curcumin ameliorate di-(2-ethylhexyl) phthalate induced testicular injury in rats [J]. General and Comparative Endocrinology, 2016, 225: 45−54. DOI: 10.1016/j.ygcen.2015.09.006
[40] DAYALAN NAIDU S, KOSTOV R V, DINKOVA-KOSTOVA A T. Transcription factors Hsf1 and Nrf2 engage in crosstalk for cytoprotection [J]. Trends in Pharmacological Sciences, 2015, 36(1): 6−14. DOI: 10.1016/j.tips.2014.10.011
-
期刊类型引用(0)
其他类型引用(1)




 下载:
下载: