Effects of Prolonged Ammonia Nitrogen Stress on Liver and Kidney Histology and Non-specific Immunity of Juvenile Carassius auratus
-
摘要:目的 研究慢性氨氮胁迫对幼鲫(Carassius auratus)肝、肾组织结构及非特异性免疫指标的影响,为研究慢性氨氮对幼鲫的危害及其集约化养殖水质管理提供理论依据。方法 以体重(3.10±0.15)g的幼鲫(C. auratus)为试验对象,通过急性毒性试验,得出96 h半致死质量浓度(LC50)和安全质量浓度(safe concentration, SC),并以此为基础,设0 mg·L−1(对照,CK)、6 mg·L−1(低质量浓度,L组)、15 mg·L−1(中质量浓度,M组)和24 mg·L−1(高质量浓度,H组)4个不同氨氮浓度处理组,试验为期60 d。分别对胁迫第15 、30 、45、60天时的幼鲫肝、肾组织的病理变化特征进行分析,并测定谷丙转氨酶(alanine aminotransferase, ALT)、谷草转氨酶(aspartate aminotransferase, AST)、过氧化氢酶(catalase, CAT)、超氧化物歧化酶(superoxide dismutase, SOD)活性变化。结果 氨氮对幼鲫的96 h半致死质量浓度LC50为289.29 mg·L−1,安全质量浓度SC为28.9 mg·L−1。在氨氮胁迫下,幼鲫组织的病理变化主要为肝细胞空泡化,核仁溶解,肝细胞轮廓模糊、排列混乱;肾小管上皮细胞水肿变性,肾小管管腔缩小,肾小球萎缩。在整个氨氮胁迫期间,肝、肾组织中各氨氮处理组的ALT、AST活性呈升高趋势,其中除15 d时L组和M组的ALT在肝脏中与对照组差异不显著(P>0.05)外,其他各时间段3个胁迫组ALT、AST活性在肝、肾组织中均与对照组差异显著(P<0.05)。肝脏中CAT、SOD比活力呈先升高后降低趋势,各胁迫组CAT比活力在45 d时与对照组差异显著,SOD比活力在15 d时与对照组差异显著。肾组织中CAT比活力在15 d时与对照组相比显著升高;各胁迫组SOD比活力在各时间段均显著高于对照组。结论 氨氮胁迫会导致幼鲫肝、肾组织严重损伤,并伤害其代谢、解毒能力和非特异性免疫能力。在集约化养殖过程中,应及时关注养殖水体中的氨氮含量,减少氨氮胁迫对鲫鱼造成的伤害,提高经济效益。Abstract:Objective Chronic ammonia nitrogen stress on juvenile crucians were studied with observations on liver and kidney histology and non-specific immunity of the fish for an improved aquaculture water quality control.Method Juvenile Carassius auratus with a body mass of (3.10±0.15) g were raised in an environmentally controlled pond. By an acute toxicity test on the fish, the semi-lethal (LC50) and safe concentrations (SC) of ammonia nitrogen over 96 h were determined. Accordingly, a control at 0 mg·L−1 and three treatments groups (i.e., low concentration L at 6 mg·L−1, medium concentration M at 15 mg·L−1, and high concentration H at 24 mg·L−1) of ammonia nitrogen in water were applied to the pond with the juvenile fish for 60 d. Histopathological changes induced by the treatments in the liver and kidney as well as the alternations occurred to the activities of alanine aminotransferase (ALT), aspartate aminotransferase (AST), catalase (CAT), and superoxide dismutase (SOD) of the fish were monitored continuously on the 15th, 30th, 45th, and 60th days.Result The acute ammonia nitrogen toxicity test on the fish showed the LC50 of 289.29 mg·L−1 and SC of 28.9 mg·L−1. The microscopic examination revealed the increasing ammonia nitrogen concentration and stress duration caused symptoms in the fish included blurred and disorganized hepatocytes with some vacuolated and cell nuclei dissolved in hepatic tissue, tumefacient renal tubular epithelial cells, narrowed renal tubular lumen, and atrophied glomerulus in nephridial tissue. The ALT and AST activities in the liver and the kidney rose with increasing ammonia nitrogen in water. The ALT in the livers of fish in L and M groups did not significantly differ from control on the 15th day (P>0.05). On the other hand, the ALT and AST in livers and kidneys of all 3 treatment groups were significantly different from control at all testing times (P<0.05). The specific activity of CAT and SOD in liver of the fish increased initially and decreased after a period of time of the treatments; that of CAT in treatment groups significantly differed from control on the 45th day; that of SOD significantly differed on the 15th day, and that of CAT in renal tissue significantly higher than that in control group on the same day; while that of SOD in all treatment groups significantly higher than that in control at all time periods.Conclusion The stress of prolonged high concentrations of ammonia nitrogen in aquacultural water caused severe damage to the liver and kidney tissues in juvenile crucians. The histopathological injuries could result in malfunctioned metabolism, compromised detoxification capacity, and reduced non-specific immunity of the fish. Consequently, for a healthy and productive aquaculture, it was deemed imperative the ammonia nitrogen content in the water be continuously monitored and strictly controlled.
-
Keywords:
- ammonia nitrogen stress /
- juvenile crucians /
- liver /
- kidney /
- non-specific immunity indices
-
0. 引言
【研究意义】鲫(Carassius auratus)是我国主要的淡水养殖鱼类,因具有营养价值高、适应性强和生长快等特点而深受消费者青睐,在我国绝大部分地区都有大规模养殖,目前鲫鱼已成为鲜活淡水鱼市场中仅次于草鱼的第二大消费品种。高密度池塘养殖在淡水鱼养殖中占据重要地位[1],该模式主要以人工投料为主,在养殖过程中会产生大量残饵和粪便,导致水质污染问题,从而引起养殖动物疾病暴发,其中氨氮是养殖水体中最主要的污染物之一。因此研究慢性氨氮胁迫对鲫幼鱼肝、肾组织及非特异性免疫指标的影响对鲫鱼养殖意义重大。【前人研究进展】氨氮是制约水生动物生长、存活和繁殖的重要环境化学因子,主要是由池塘或海洋黏土沉积物的咸水渗透[2]、田地施肥、大气沉降、农田固氮[3]、粪便残饵等产生,因此养殖期间水体管理不当会造成氨氮的快速产生和积累。当水生环境中的氨氮浓度过高时,会导致鱼类氨氮生物蓄积,引起酶代谢紊乱,从而过于兴奋,降低免疫力,损害鱼类的中枢神经系统等[4],并对鱼类的存活、生长代谢、组织结构、生理和免疫功能等产生毒性效应[5]。近年来,国内外学者在氨氮对鱼类养殖的影响方面做了大量研究,结果表明,急性氨氮胁迫会引起大口黑鲈(Micropterus salmoides)肝脏氧化应激[6],洞庭青鲫(Carassius auratus indigentiaus)幼鱼抗氧化防御系统功能紊乱[7],进入鱼体血液循环快速引发毒性效应[8],导致斑石鲷(Oplegnathus punctatus)幼鱼行为异常[9]等。慢性氨氮胁迫会抑制幼年钝吻鲷(Megalobrama amblycephala)的生长[10],诱发黄颡鱼(Pelteobagrus fulvidraco)鳃出现严重的增生和炎症[11],导致尼罗罗非鱼(Oreochromis niloticus)[12]和团头鲂(Megalobrama Amblycephala)[13]发生氧化应激和免疫抑制效应,肝脏出现不同程度的损伤。【本研究切入点】目前,虽然关于氨氮胁迫对鱼类影响的研究有很多,但都集中在急性胁迫方面,有关慢性氨氮胁迫的研究较少,且影响指标大部分集中在生长方面,对鱼类肝、肾组织及非特异性免疫指标的影响鲜少报道。【拟解决的关键问题】本研究通过研究慢性氨氮胁迫对鲫幼鱼肝、肾组织及非特异性免疫指标的影响,以期为解析氨氮对鲫鱼的危害及鲫鱼集约化养殖过程中水体氨氮的调控、水质管理提供科学的理论依据。
1. 材料与方法
1.1 试验动物及管理
试验动物为从太原鱼种场购买的健康幼鲫,体长(3.5±0.2) cm,体重(3.10±0.15) g,共计500尾。试验用水为充分曝气后的自来水,水温控制在(25±1) ℃,pH为(7.5±0.3),溶解氧不低于5 mg·L−1,每天13:00换全水1次,并重新加入配制好的氨氮溶液,换水1 h后投喂饲料,正式试验前停食1 d。
1.2 试验方法
1.2.1 急性毒理试验
暂养结束后开始进行急性毒性试验,以NH4Cl配制质量浓度为10 g·L−1的NH4Cl母液,根据需要稀释成不同质量浓度的使用液,选取300尾幼鲫,将试验鱼饲养于NH4Cl质量浓度分别为240、320、400、480、560 mg·L−1的5个处理组中,同时以未添加氨氮的正常饲养用水作为对照组,每组设置5个重复,每个重复10尾鱼。试验期间不投饵,分别记录24、48、72、96 h的个体死亡数,并及时清除死亡个体,以停止呼吸、针刺无反应作为死亡标准。每天换水后对氨氮浓度进行校正,使用奥克丹多参数水质分析仪进行氨氮浓度检测,以确保试验水体中氨氮浓度与设计浓度吻合。
以Bliss法求取不同攻毒时间的96 h半致死质量浓度(LC50),同时获得不同胁迫时间的回归方程、可决系数和95%置信区间,并估算出氨氮对鲫鱼幼鱼的和安全质量浓度(safe concentration, SC)。安全质量浓度的计算公式为:
SC/(mg·L−1) =0.1×96 h LC50
1.2.2 慢性毒理试验
通过预试验确定体重为3.10 g左右的幼鲫96 h半致死浓度LC50为289.29 mg·L−1,安全浓度(SC)按照LC50的10%估算为28.9 mg·L−1。试验于60 cm×40 cm×40 cm的养殖箱中进行,将试验鱼随机分为4组,设0 mg·L−1(对照,CK)、6 mg·L−1(低质量浓度,L组)、15 mg·L−1(中质量浓度,M组)和24 mg·L−1(高质量浓度,H组)4个氨氮浓度梯度处理,每组设置5个重复,每个重复10尾鱼。试验期间维持试验水体的溶氧量和温度,每天13: 00全换水1次。所换水均经过24 h的曝气,换水后再重新加入配置好的氨氮溶液,使用奥克丹多参数水质分析仪进行氨氮浓度检测,确保试验水体中氨氮浓度与设计浓度吻合。每组浓度实测值分别为(0.40±0.02)、(6.05±0.21)、(15.06±0.10)、(24.05±0.14)mg·L−1。参考《地面水环境质量标准非离子氨换算方法》[14]将其转换成非离子氨浓度,分别为0.00、0.04、0.10、0.16 mg·L−1。
1.2.3 样品采集
分别于第15、30、45、60天从各组随机取10尾试验鱼进行解剖取样,采集其肝脏、肾脏组织,置于有波恩氏液(Bouin’s liquid)的1.5 mL离心管中固定,用于制作石蜡切片。每组另取5尾鱼的肝、肾组织样品置于−80 ℃冰箱保存,用于非特异性免疫指标的测定。
1.2.4 幼鲫肝、肾组织结构分析
用自来水冲洗已经固定好的组织12 h,采用常规石蜡切片制作方法,经酒精(50%、75%、85%、95%、100%、100%)脱水后,用1/2二甲苯(V二甲苯∶V酒精=1∶1)、二甲苯Ⅰ、二甲苯Ⅱ透明,之后用1/2石蜡(V石蜡∶V二甲苯=1∶1)、经石蜡Ⅰ、Ⅱ、Ⅲ依次浸蜡,再进行包埋、切片、展片、脱蜡、H.E.染色,最后中性树脂封片,自然风干后置于光学显微镜下观察并拍照。
1.2.5 酶活性测定
使用预冷过的PBS将分离的肝脏、肾脏组织充分匀浆,
3000 r·min−1冷冻离心20 min后取上清液,用于测定肝、肾组织的谷丙转氨酶(Alanine aminotransferase, ALT)、谷草转氨酶(Aspartate aminotransferase, AST)、过氧化氢酶(Catalase, CAT)和超氧化物歧化酶(Superoxide dismutase, SOD)活性。参照《水产动物机能学实验》采用比色法测定肝、肾组织中ALT、AST活性。以37 ℃与底物作用30 min、产生2.5 μg的丙酮酸为一个ALT活力单位(U·mL−1)。在37 ℃下每mL组织匀浆样本每分钟催化产生1 nmol丙酮酸定义为一个AST活力单位(U·mL−1)。用南京建成生物公司研究所研制的试剂盒测定CAT,按说明书操作。CAT活力定义为每毫克组织蛋白每秒钟分解1 µmol的H2O2为一个活力单位(U·mg−1)。使用南京建成生物公司研究所研制的试剂盒测定SOD,按说明书操作。SOD活力定义为每毫克组织蛋白在1 mL反应液中SOD抑制率达50%时所对应的SOD量为一个SOD活力单位(U·mg−1)。1.3 数据处理
试验数据用SPSS 21.0的单因素(ANOVA)方法进行分析,数据结果以平均值±标准差(Mean ± SD)表示,采用Duncan法进行多重比较,显著水平为P<0.05,极显著水平为P<0.01。
2. 结果与分析
2.1 氨氮对幼鲫的半致死浓度(LC50)及安全浓度
氨氮对幼鲫的急性毒性测定结果见表1。由表1可知,幼鲫对氨氮敏感,随着处理时间的延长,半致死质量浓度值呈减小趋势,96 h LC50为289.29 mg·L−1,转换成非离子氨为1.98 mg·L−1,SC按照LC50的10%估算为28.9 mg·L−1,转换成非离子氨为0.20 mg·L−1。
表 1 氨氮对鲫幼鱼急性毒性试验结果Table 1. Acute toxicity test of ammonia nitrogen on mortality of juvenile C. auratus胁迫时间
Stress time/h回归方程
Regressive equation相关系数R2 半致死浓度
LC50/(mg·L−1)95%置信区间
95% confidence interval/(mg·L−1)安全浓度SC/
(mg·L−1)24 P= 5.7327 X-15.0845 0.9939 427.86 393.45~465.28 42.8 48 P= 7.7711 X-19.9359 0.9985 367.62 356.67~378.91 36.8 72 P= 8.6415 X-21.7653 0.9879 330.14 271.14~401.98 33.0 96 P= 5.5711 X-13.7124 0.9980 289.29 264.08~316.90 28.9 LC50采用概率单位法计算;P为死亡率;X为NH4+质量浓度对数。
LC50 was calculated by probabilistic unit method; P is mortality probability unit; X is log of NH4+ concentration.2.2 慢性氨氮胁迫对幼鲫肝组织的影响
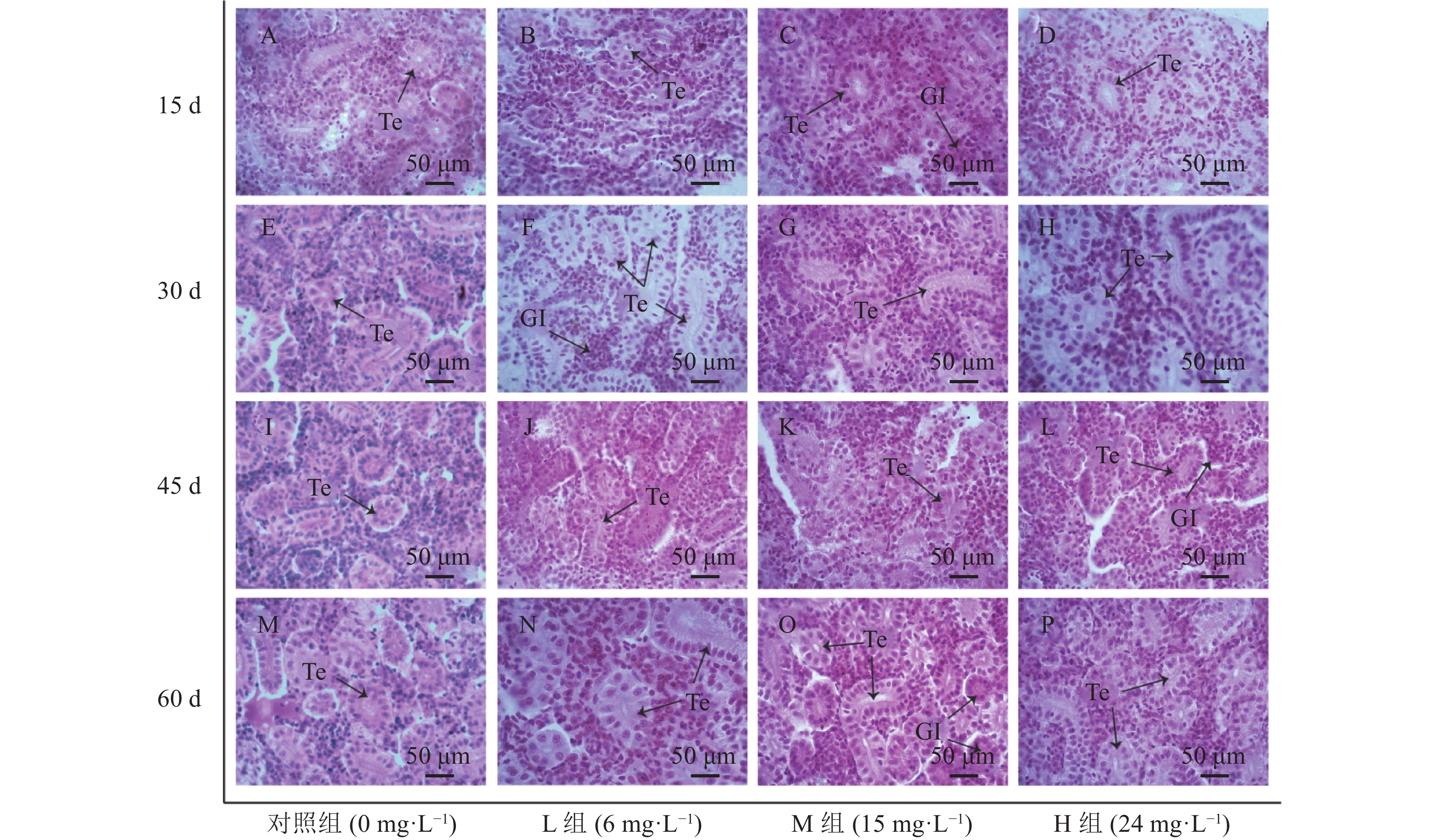
慢性氨氮胁迫对幼鲫肝组织结构的影响见图1。对照组肝脏组织结构完整,肝细胞排列整齐,轮廓清晰,核仁明显,肝血窦大小正常,清晰可见(图1A、E、I、M)。
L组(6 mg·L−1)前两个周期内,肝细胞无明显变化,细胞排列整齐,细胞轮廓清晰、大小正常,细胞核明显(图1B、F)。胁迫45 d,肝组织部分细胞开始出现空泡化,部分细胞核核仁溶解(图1J);胁迫60 d,肝细胞空泡化严重,胞浆开始变得透明,各个细胞间细胞膜界限不明显(图1N)。
M组(15 mg·L−1)胁迫15 d时,肝细胞无明显变化(图1C)。胁迫30 d,肝细胞肿大,核仁出现溶解现象(图1G)。胁迫45 d,肝细胞轮廓模糊、排列混乱、水肿变性严重,部分细胞核核仁溶解,肝血窦扩张(图1K)。胁迫60 d,肝细胞空泡变性,核仁溶解(图1O)。
H组(24 mg·L−1)胁迫15 d,观察到肝细胞肿胀,细胞核发生偏移,核仁溶解,细胞轮廓模糊,排列混乱(图1D)。胁迫45 d,肝细胞水肿变性、空泡化严重,多数细胞核核仁溶解(图1L)。胁迫60 d,肝细胞水肿变性严重,大量细胞核溶解,轮廓严重模糊,肝组织结构严重损伤(图1P)。
2.3 慢性氨氮胁迫对幼鲫肾组织的影响
慢性氨氮胁迫对幼鲫肾组织的影响见图2。对照组肾组织结构整齐(图2A、E、I、M),肾小管上皮细胞结构完整,肾小球饱满。L组(6 mg·L−1)前两个周期内,肾组织细胞无明显变化(图2B、F),胁迫45 、60 d时后肾上皮细胞水肿变性,肾小球萎缩(图2J、N)。M组(15 mg·L−1)胁迫第15天时无明显变化(图2C),第30 d、45 d和60 d观察到肾小管上皮细胞肿胀变性,肾小管胞浆疏松透明(图2G、K、O)。H组(24 mg·L−1)各时间段均出现肾上皮细胞水样变性,肾小管管腔缩小(图2D、H、L、P)。
2.4 慢性氨氮胁迫对幼鲫肝、肾脏非特异性免疫指标的影响
2.4.1 慢性氨氮对幼鲫肝、肾脏ALT的影响
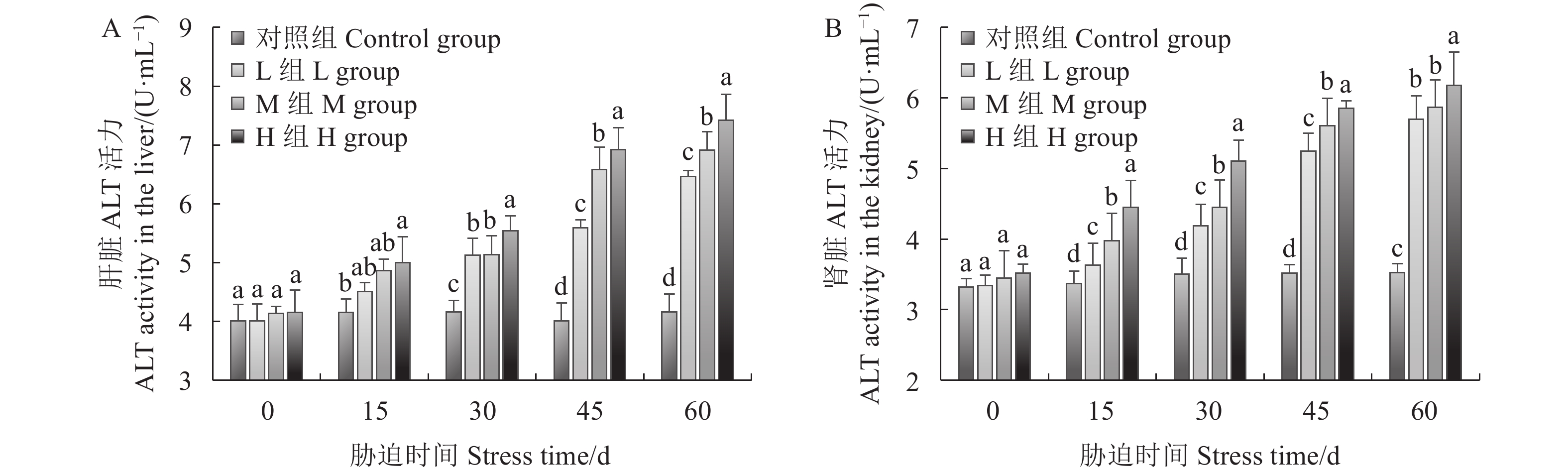
如图3A所示,幼鲫经氨氮胁迫后,肝脏中ALT活力随着氨氮浓度的升高以及氨氮暴露时间的增加而增强。15 d时,H组(24 mg·L−1)ALT活力显著高于对照组,30 ~ 60 d,各胁迫组在肝组织中ALT活力均显著高于对照组。30 d时,L组(6 mg·L−1)和M组(15 mg·L−1)与H组(24 mg·L−1)差异显著,45 ~ 60 d,各胁迫组之间差异显著。如图3B所示,随着氨氮浓度的升高和暴露时间的延长,肾脏ALT的活性一直保持升高的趋势。15 ~ 60 d各胁迫组肾脏ALT的活力均与对照组差异显著。15~45 d各胁迫组之间差异显著,60 d时L组、M组与H组差异显著。
2.4.2 慢性氨氮对幼鲫肝、肾脏AST的影响
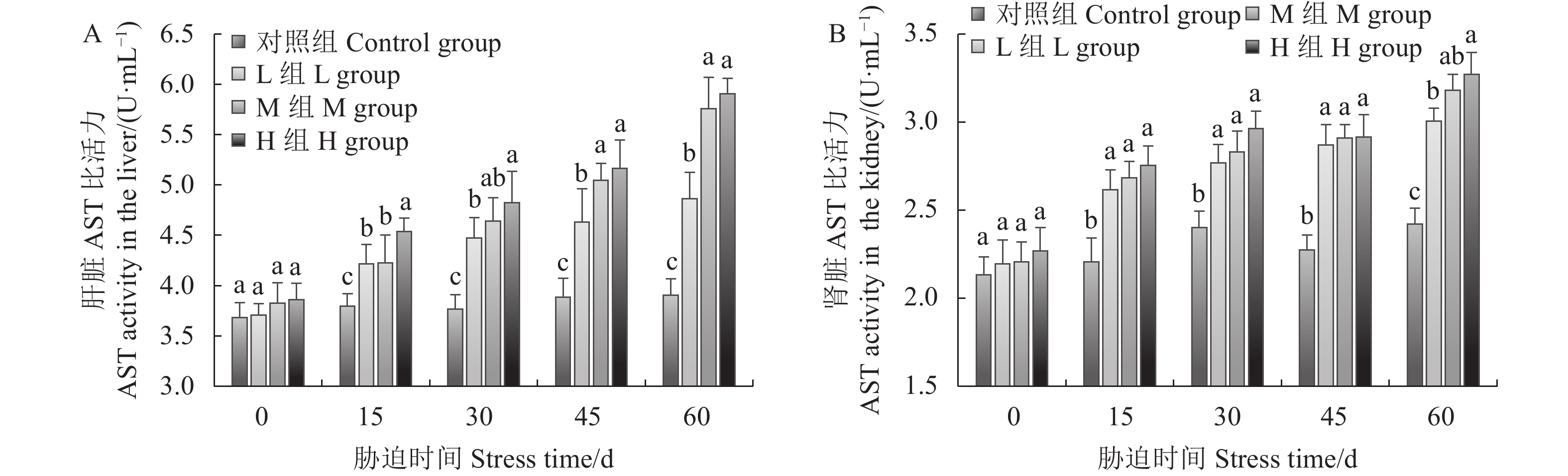
由图4可知,在整个氨氮胁迫期间,随着氨氮浓度的升高和暴露时间的延长,3个试验组中肝、肾组织AST活性升高且与对照组差异显著;肝组织在15 ~ 30 d时,L组与M组AST活性无显著差异,第15 d时二者与H组差异显著,肾组织在60 d时,L组与H组之间AST活性差异显著。
2.4.3 慢性氨氮对幼鲫肝、肾脏CAT的影响
由图5A可知,幼鲫经氨氮胁迫后,其肝组织中CAT的活性随着氨氮浓度的升高以及氨氮暴露时间的增加逐渐升高,于45 d时达到峰值,L组、M组、H组分别为372.89 、379.30、381.26 U·mg−1,且3组均与对照组差异显著;60 d时各试验组CAT活性开始降低,但仍与对照组差异显著。
由图5B可知,随着氨氮浓度的升高,各试验组在15 d时肾组织CAT与对照组相比有升高趋势,且差异显著。L组和M组在60 d时肾组织CAT比活力达到峰值,分别为230.65、279.78 U·mg−1。H组肾组织CAT比活力在45 d时达到峰值,60 d时较之前比活力有下降趋势。
2.4.4 慢性氨氮对幼鲫肝、肾脏SOD的影响
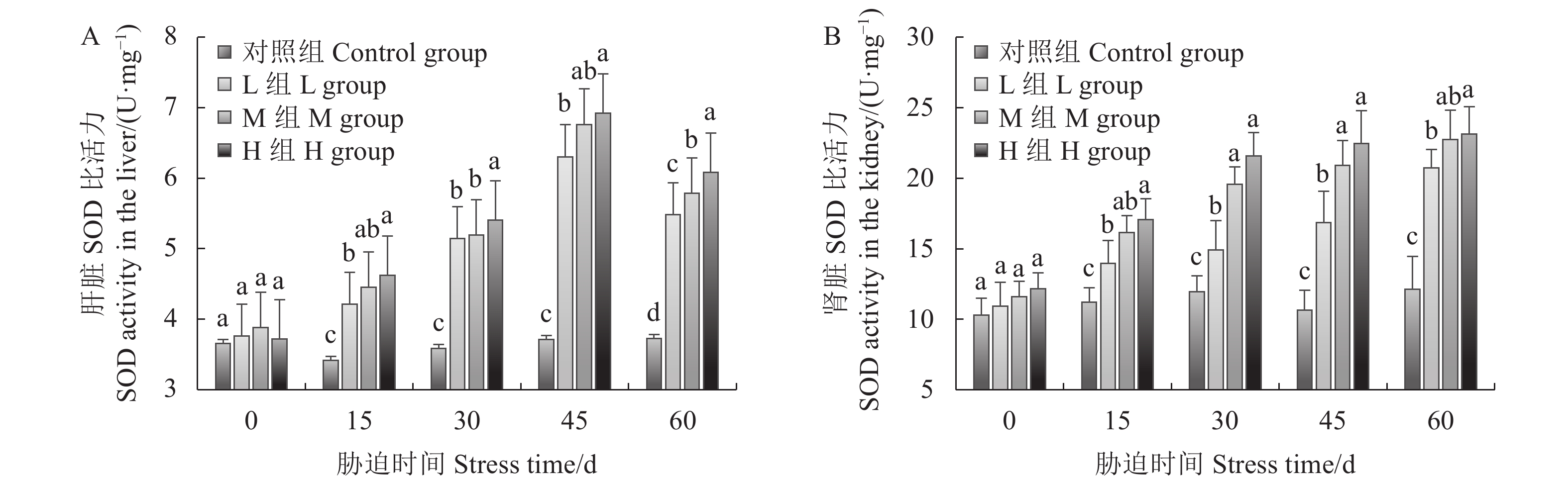
由图6A可知,幼鲫经氨氮胁迫后,其肝脏中SOD的活性随着氨氮浓度的升高以及氨氮暴露时间的增加而增加,各氨氮浓度胁迫15 d时肝组织中SOD比活力就已经显著高于对照组,在胁迫45 d时达到峰值,L组、M组、H组分别为6.31、6.76、6.93 U·mg−1。60 d时各胁迫组肝脏SOD的活性有降低的趋势,但仍与对照组存在显著差异。
由图6B可知,随着氨氮浓度的升高,各试验组肾组织SOD活性升高。各胁迫组在各时间段SOD酶的比活力均显著高于对照组。15 、60 d时,L组与H组之间差异显著,30 、45 d时,L组与M和H两组之间差异显著。
3. 讨论
3.1 氨氮胁迫对幼鲫的急性毒性效应
养殖水体中氨氮对鱼类的致毒作用主要是由于水体中非离子氨浓度过高,表现为不同程度的组织损伤[15]。非离子氨对水生生物危害较大的主要原因是非离子氨是带有较强脂溶性的中性分子,它可以透过细胞膜进入水生生物体,影响水生生物体内的酶水解反应进程,对细胞膜稳定性也会产生影响,导致水生生物的某些生理生化指标与生长指标发生改变,引起水生生物发病,严重时致死[16]。
本研究以幼鲫为试验对象进行96 h的急性氨氮攻毒,通过Bliss法得出其LC50为289.29 mg·L−1,SC按照LC50的10%估算,为28.9 mg·L−1。由于不同种类水生生物对氨氮胁迫的耐受力不同,因此受氨氮胁迫后的致死率也不相同。唐忠林等[17]在对大口黑鲈(M. salmoides)北方亚种引进种F1代幼鱼的急性氨氮胁迫研究中得到96 h半致死浓度和安全浓度分别为25.08 、2.51 mg·L−1;杨西伟等[18]在研究刀鲚(Coilia nasus)幼鱼的急性毒性时得到其96 h半致死浓度和安全浓度分别为46.83 mg·L−1和4.68 mg·L−1。幼鲫对于氨氮的毒性耐受力要高于大多数硬骨鱼的幼鱼[14, 15],也远高于经济虾蟹类[19 - 21],但低于海湾豹蟾鱼(Opsanus beta)、毒棘豹蟾鱼(Opsanus tau)和斑光蟾鱼(Porichthys notatus),因其具有通过尿素合成进行氨解毒的替代或附加机制[22]。
3.2 慢性氨氮胁迫对幼鲫肝、肾组织的影响
肝脏是鱼体内最大的消化腺,其基本组织结构能很大程度上揭示鱼类的营养需求及生态习性的内部本质,在新陈代谢、解毒和免疫中都起着重要作用[23]。研究表明,鱼体暴露在高浓度氨氮的水体中,会造成肝组织的损伤,导致其肝脏出现水肿、空泡化严重和局部坏死等病变[24 - 26]。本研究中,肝脏出现肿大、空泡化、细胞核溶解、细胞轮廓不清晰等现象,且随着氨氮浓度的升高及胁迫时间的延长,肝组织损伤逐渐严重,这与其他鱼类中的研究结果一致。
肾脏是鱼类的排泄器官,鱼类的代谢产物主要通过肾脏排出体外[27]。研究表明,外界水环境中的污染物如农药[28]、重金属[29]等均会引起鱼类肾脏的病变。本研究中,肾组织在氨氮的胁迫下,出现肾小管管腔缩小、肾小管上皮细胞水肿变性、肾小球萎缩等现象,且随着氨氮浓度的升高及胁迫时间的延长,肾组织损伤逐渐严重。在急性氨胁迫对团头鲂(Megalobrama amblycephala)肾组织结构影响的研究中,肾脏出现同样的变化[30]。因此,高浓度的氨氮或长时间的氨氮胁迫都会导致鱼类肾脏出现损伤。
3.3 慢性氨氮胁迫对幼鲫肝、肾非特异性免疫指标的影响
氨氮暴露会导致水产养殖动物免疫功能受到抑制[10, 31, 32]。鱼类的非特异性免疫在应对环境应激中起主导作用,其中免疫酶主要包括ALT、AST、CAT、SOD等[33, 34]。本研究发现,氨氮胁迫对幼鲫肝、肾组织的非特异性免疫指标影响显著,随着胁迫时间的延长,不同氨氮浓度下幼鲫ALT、AST、CAT和SOD活性均发生了明显变化。
AST与ALT是一种重要的氨基转移酶,广泛存在于生物机体内,对生物机体内的氨基酸合成以及分解代谢具有重要作用,是评价肝肾损伤的重要指标[35, 36]。当组织中氨浓度升高时,会导致膜转运能力降低从而减少氧的摄入,使得机体能量消耗增加,诱发机体生理紊乱,损害水生动物的健康[37]。据Chandra等[38]研究发现,AST由肝细胞合成,通常见于肝脏,而ALT常见于肝脏和肾脏中,因此AST和ALT是肝功能检查中的标志物。Rahimnejad等[39]试验表明,当肝细胞受损时,ALT和AST比活力增强。EL Euony等[40]研究显示,革胡子鲶(Clarias garipenus)噻虫嗪中毒后,其肝脏和头肾出现退行性改变和坏死,AST、ALT活性水平显著升高。本试验中各浓度组肝、肾组织中AST和ALT的比活力随着氨氮暴露时间的延长而明显升高,且一直保持较高水平,说明高浓度的氨氮或者氨氮长期暴露会导致鱼类肝、肾受到损伤。
CAT和SOD是机体抗氧化系统中最重要的两种抗氧化酶,主要作用是清除机体产生的氧自由基(ROS),防止由于ROS过多造成机体损伤[41]。本研究发现,幼鲫肝组织中CAT和SOD的活性呈现先升后降的趋势,且均与对照组有极显著差异,这与姜会民[42]氨氮胁迫鲤(Cyprinus carpio)幼鱼的结果相似。其主要原因是在受到氨氮胁迫时,鱼体产生了毒物兴奋效应,机体产生大量的ROS,导致机体损伤,之后机体启动抗氧化防御系统抑制ROS的生产,使得CAT、SOD活性呈现升高趋势,但经过高浓度且长时间的氨氮暴露会造成肝组织损伤,导致组织中2种抗氧化酶的活性降低。氨氮胁迫后肾组织中的SOD和CAT活性也明显高于对照组,这是为了清除过多的ROS,以减轻过氧化损伤。而高浓度组(H组)CAT活性在60 d时降低,这可能是因为长时间高浓度胁迫所产生的有害物质超过了鱼体的耐受上限。
4. 结论
氨氮对幼鲫的96 h LC50为289.29 mg·L−1,安全浓度为28.9 mg·L−1,随着氨氮浓度的升高和胁迫时间的延长,幼鲫肝和肾组织受到的损害增加。慢性氨氮的毒性会对幼鲫的肝、肾组织产生伤害,并严重影响其非特异性免疫指标,进而影响鱼体健康。因此在进行鲫鱼养殖时,应关注水体的氨氮浓度,防止氨氮对鲫鱼的毒性效应。
-
图 3 慢性氨氮胁迫对幼鲫肝、肾ALT的影响
不同小写字母表示同一胁迫时间不同处理组间差异显著(P<0.05)。图4、5、6同。
Figure 3. Effect of chronic ammonia nitrogen stress on ALT in livers and kidneys of juvenile C. auratus
Data with different lowercase letters indicate significant difference among treatment groups in same duration under stress at P<0.05. Same for Figs.4, 5 and 6.
表 1 氨氮对鲫幼鱼急性毒性试验结果
Table 1 Acute toxicity test of ammonia nitrogen on mortality of juvenile C. auratus
胁迫时间
Stress time/h回归方程
Regressive equation相关系数R2 半致死浓度
LC50/(mg·L−1)95%置信区间
95% confidence interval/(mg·L−1)安全浓度SC/
(mg·L−1)24 P= 5.7327 X-15.0845 0.9939 427.86 393.45~465.28 42.8 48 P= 7.7711 X-19.9359 0.9985 367.62 356.67~378.91 36.8 72 P= 8.6415 X-21.7653 0.9879 330.14 271.14~401.98 33.0 96 P= 5.5711 X-13.7124 0.9980 289.29 264.08~316.90 28.9 LC50采用概率单位法计算;P为死亡率;X为NH4+质量浓度对数。
LC50 was calculated by probabilistic unit method; P is mortality probability unit; X is log of NH4+ concentration. -
[1] SUBASINGHE R, SOTO D, JIA J S. Global aquaculture and its role in sustainable development [J]. Reviews in Aquaculture, 2009, 1(1): 2−9. DOI: 10.1111/j.1753-5131.2008.01002.x
[2] RUSYDI A F, ONODERA S I, SAITO M, et al. Potential sources of ammonium-nitrogen in the coastal groundwater determined from a combined analysis of nitrogen isotope, biological and geological parameters, and land use [J]. Water, 2020, 13(1): 25. DOI: 10.3390/w13010025
[3] ZHANG W S, SWANEY D P, LI X Y, et al. Anthropogenic point-source and non-point-source nitrogen inputs into Huai River Basin and their impacts on riverine ammonia–nitrogen flux [J]. Biogeosciences, 2015, 12(14): 4275−4289. DOI: 10.5194/bg-12-4275-2015
[4] XU Z K, CAO J, QIN X M, et al. Toxic effects on bioaccumulation, hematological parameters, oxidative stress, immune responses and tissue structure in fish exposed to ammonia nitrogen: A review [J]. Animals: an Open Access Journal from MDPI, 2021, 11(11): 3304.
[5] 熊小琴, 王岚, 史庆超, 等. 氨氮对鱼类的毒性效应研究进展 [J]. 贵州农业科学, 2021, 49(7):81−87. XIONG X Q, WANG L, SHI Q C, et al. Research progress on toxic effects of ammonia nitrogen on fish [J]. Guizhou Agricultural Sciences, 2021, 49(7): 81−87. (in Chinese)
[6] 刘品, 王文博, 李超, 等. 急性氨氮胁迫下大口黑鲈的肝脏转录组特征分析 [J]. 水生生物学报, 2024, 48(5):713−724. LIU P, WANG W B, LI C, et al. Liver transcriptome of largemouth bass (Micropterus samoides) under acute ammonia nitrogen stress [J]. Acta Hydrobiologica Sinica, 2024, 48(5): 713−724. (in Chinese)
[7] 刘可欣, 林旺, 罗惠敏, 等. 氨氮胁迫对青鲫幼鱼急性毒性和抗氧化功能的影响 [J]. 湖南文理学院学报(自然科学版), 2023, 35(2):57−61,87. LIU K X, LIN W, LUO H M, et al. Effects of ammonia nitrogen stress on acute toxicity and antioxidant capacity of juvenile Dongtingking crucian carp (Carassius auratus indigentiaus ) [J]. Journal of Hunan University of Arts and Science (Science and Technology), 2023, 35(2): 57−61,87. (in Chinese)
[8] 高云涛, 高云红, 王嘉伟, 等. 氨氮急性胁迫对许氏平鲉血液生化指标影响 [J]. 海洋科学, 2023, 47(8):49−59. GAO Y T, GAO Y H, WANG J W, et al. Effect of acute ammonia stress on the plasma biochemical indexes of Sebastes schlegelii [J]. Marine Sciences, 2023, 47(8): 49−59. (in Chinese)
[9] 张盛坤, 徐进虎, 孙雪倩, 等. 氨氮急性胁迫对斑石鲷幼鱼行为模式和血液学参数的影响 [J]. 中国海洋大学学报(自然科学版), 2023, 53(3):120−132. ZHANG S K, XU J H, SUN X Q, et al. Effects of acute ammonia nitrogen stress on behavior pattern and hematological parameters of juvenile spotted knifejaw (Oplegnathus punctatus) [J]. Periodical of Ocean University of China, 2023, 53(3): 120−132. (in Chinese)
[10] ZHANG W X, XIA S L, ZHU J, et al. Growth performance, physiological response and histology changes of juvenile blunt snout bream, Megalobrama amblycephala exposed to chronic ammonia [J]. Aquaculture, 2019, 506: 424−436. DOI: 10.1016/j.aquaculture.2019.03.072
[11] ZHONG L, LIU S, ZUO F Y, et al. The IL17 signaling pathway: A potential signaling pathway mediating gill hyperplasia and inflammation under ammonia nitrogen stress was identified by multi-omics analysis [J]. Science of the Total Environment, 2023, 867: 161581. DOI: 10.1016/j.scitotenv.2023.161581
[12] ESAM F, KHALAFALLA M M, GEWAILY M S, et al. Acute ammonia exposure combined with heat stress impaired the histological features of gills and liver tissues and the expression responses of immune and antioxidative related genes in Nile tilapia [J]. Ecotoxicology and Environmental Safety, 2022, 231: 113187. DOI: 10.1016/j.ecoenv.2022.113187
[13] GUO H H, LIN W, WU X Y, et al. Survival strategies of Wuchang bream (Megalobrama amblycephala) juveniles for chronic ammonia exposure: Antioxidant defense and the synthesis of urea and glutamine[J]. Comparative Biochemistry and Physiology Part C: Toxicology & Pharmacology, 2020, 230: 108707.
[14] 国家环境保护局科技标准司标准处. 地面水环境质量标准非离子氨换算方法[J]. 中国环境监测, 1995, 11(4): 9−21. Standards Department of the Science and Technology Standards Department of the National Environmental Protection Administration. Conversion method of nonionic ammonia in surface water environmental quality standard[J]. Environmental Monitoring in China, 1995, 11(4): 9−21. (in Chinese)
[15] HAWKES J W. The effects of xenobiotics on fish tissues: Morphological studies [J]. Federation Proceedings, 1980, 39(14): 3230−3236.
[16] 雷衍之. 养殖水环境化学[M]. 北京: 中国农业出版社, 2004. [17] 唐忠林, 张佳佳, 周国勤, 等. 氨氮对大口黑鲈北方亚种引进种F1代幼鱼急性毒性及生理变化的影响 [J]. 淡水渔业, 2023, 53(5):97−103. TANG Z L, ZHANG J J, ZHOU G Q, et al. Effects of ammonia nitrogen on acute toxicity and physiological changes for introduction of Micropterus salmoides F1 juvenile [J]. Freshwater Fisheries, 2023, 53(5): 97−103. (in Chinese)
[18] 杨西伟, 徐钢春, 高俊, 等. 氨氮胁迫对刀鲚稚、幼鱼的急性毒性及抗氧化酶的影响 [J]. 海洋湖沼通报, 2021, 43(4):127−132. YANG X W, XU G C, GAO J, et al. Effects of ammonia nitrogen stress on acute toxicity and antioxidant enzymes of Coilia nasus [J]. Transactions of Oceanology and Limnology, 2021, 43(4): 127−132. (in Chinese)
[19] 吴乐, 李嘉尧, 周文宗, 等. 氨氮短期胁迫与恢复对克氏原螯虾的影响 [J]. 水产科学, 2024, 43(3):390−399. WU L, LI J Y, ZHOU W Z, et al. Effects of short-term ammonia nitrogen stress and recovery on red swamp crayfish Procambarus clarkii [J]. Fisheries Science, 2024, 43(3): 390−399. (in Chinese)
[20] WANG T, LI W H, SHAN H W, et al. Responses of energy homeostasis and lipid metabolism in Penaeus vannamei exposed to ammonia stress [J]. Aquaculture, 2021, 544: 737092. DOI: 10.1016/j.aquaculture.2021.737092
[21] 彭军辉, 陈丽英, 程长洪, 等. 氨氮对拟穴青蟹的急性毒性及对其血清免疫相关酶活力的影响 [J]. 渔业科学进展, 2018, 39(5):114−121. PENG J H, CHEN L Y, CHENG C H, et al. Acute toxicity of ammonia nitrogen to Scylla paramamosain and its influence on immune factors in serum [J]. Progress in Fishery Sciences, 2018, 39(5): 114−121. (in Chinese)
[22] WANG Y X, WALSH P J. High ammonia tolerance in fishes of the family Batrachoididae (Toadfish and Midshipmen) [J]. Aquatic Toxicology, 2000, 50(3): 205−219. DOI: 10.1016/S0166-445X(99)00101-0
[23] 刘思雨. 鱼类肝脏组织形态的影响因素探讨 [J]. 南方农业, 2022, 16(14):175−177. LIU S Y. Study on the influencing factors of liver morphology in fish [J]. South China Agriculture, 2022, 16(14): 175−177. (in Chinese)
[24] ZOU J H, HU P, WANG M Y, et al. Liver injury and metabolic dysregulation in largemouth bass (Micropterus salmoides) after ammonia exposure [J]. Metabolites, 2023, 13(2): 274. DOI: 10.3390/metabo13020274
[25] 吴利敏, 徐瑜凤, 李永婧, 等. 急性氨氮胁迫对淇河鲫幼鱼脑、鳃、肝、肾组织结构的影响 [J]. 中国水产科学, 2020, 27(7):789−800. WU L M, XU Y F, LI Y J, et al. Effects of acute ammonia nitrogen exposure on brain, gill, liver, and kidney histology of Qi River crucian carp(Carassius auratus) [J]. Journal of Fishery Sciences of China, 2020, 27(7): 789−800. (in Chinese)
[26] GUO M J, YAN Q, DONG Y X, et al. Apoptotic changes, oxidative stress and immunomodulatory effects in the liver of Japanese seabass (Lateolabrax japonicus) induced by ammonia-nitrogen stress during keep-live transport [J]. Biology, 2023, 12(6): 769. DOI: 10.3390/biology12060769
[27] BJØRGEN H, KOPPANG E O. Anatomy of teleost fish immune structures and organs [J]. Immunogenetics, 2021, 73(1): 53−63. DOI: 10.1007/s00251-020-01196-0
[28] LACY B, RAHMAN M S. Interactive effects of high temperature and pesticide exposure on oxidative status, apoptosis, and renin expression in kidney of goldfish: Molecular and cellular mechanisms of widespread kidney damage and renin attenuation [J]. Journal of Applied Toxicology: JAT, 2022, 42(11): 1787−1806. DOI: 10.1002/jat.4357
[29] HERMENEAN A, DAMACHE G, ALBU P, et al. Histopatological alterations and oxidative stress in liver and kidney of Leuciscus cephalus following exposure to heavy metals in the Tur River, North Western Romania [J]. Ecotoxicology and Environmental Safety, 2015, 119: 198−205. DOI: 10.1016/j.ecoenv.2015.05.029
[30] 张武肖, 孙盛明, 戈贤平, 等. 急性氨氮胁迫及毒后恢复对团头鲂幼鱼鳃、肝和肾组织结构的影响 [J]. 水产学报, 2015, 39(2):233−244. ZHANG W X, SUN S M, GE X P, et al. Acute effects of ammonia exposure on histopathology of gill, liver and kidney in juvenile Megalobrama amblycephala and the post-exposure recovery [J]. Journal of Fisheries of China, 2015, 39(2): 233−244. (in Chinese)
[31] CHENG C H, YANG F F, LING R Z, et al. Effects of ammonia exposure on apoptosis, oxidative stress and immune response in pufferfish (Takifugu obscurus) [J]. Aquatic Toxicology, 2015, 164: 61−71. DOI: 10.1016/j.aquatox.2015.04.004
[32] LI M, GONG S Y, LI Q, et al. Ammonia toxicity induces glutamine accumulation, oxidative stress and immunosuppression in juvenile yellow catfish Pelteobagrus fulvidraco[J]. Comparative Biochemistry and Physiology Part C: Toxicology & Pharmacology, 2016, 183: 1-6.
[33] 赵斌, 周红学, 李成林, 等. 氨氮胁迫对刺参“鲁海1号” 非特异性免疫的影响 [J]. 西北农林科技大学学报(自然科学版), 2022, 50(2):17−24. ZHAO B, ZHOU H X, LI C L, et al. Effects of ammonia nitrogen stress on activities of non-specific immune enzymes of sea cucumber Luhai No. 1 [J]. Journal of Northwest A & F University (Natural Science Edition), 2022, 50(2): 17−24. (in Chinese)
[34] NAYAK S, DAS S, KUMAR R, et al. Biochemical and histopathological alterations in freshwater fish, Labeo rohita (Hamilton, 1822) upon chronic exposure to a commonly used hopper insecticide, triflumezopyrim [J]. Chemosphere, 2023, 337: 139128. DOI: 10.1016/j.chemosphere.2023.139128
[35] 班同, 尹晶, 吴垠. 氨氮浓度对工厂化养殖漠斑牙鲆血液指标的影响 [J]. 河北渔业, 2013, (8):1−5. DOI: 10.3969/j.issn.1004-6755.2013.08.001 BAN T, YIN J, WU Y. Effect of ammonia nitrogen concentration on blood indexes of industrial cultured PARALICHTHYS olivaceus [J]. Hebei Fisheries, 2013(8): 1−5. (in Chinese) DOI: 10.3969/j.issn.1004-6755.2013.08.001
[36] ORTIZ G G, BITZER-QUINTERO O K, ZÁRATE C B, et al. Monosodium glutamate-induced damage in liver and kidney: A morphological and biochemical approach [J]. Biomedicine & Pharmacotherapy, 2006, 60(2): 86−91.
[37] TAHERI MIRGHAED A, FAYAZ S, HOSEINI S M. Effects of dietary 1, 8-cineole supplementation on serum stress and antioxidant markers of common carp (Cyprinus carpio) acutely exposed to ambient ammonia [J]. Aquaculture, 2019, 509: 8−15. DOI: 10.1016/j.aquaculture.2019.04.071
[38] CHANDRA S J, MAHADIMANE P V, SAQIB A, et al. Barium Chloride impairs physiology and brain glutamate in Cirrhinus mrigala during a short period of interaction [J]. Egyptian Journal of Aquatic Biology and Fisheries, 2020, 24(7): 995−1003. DOI: 10.21608/ejabf.2020.144745
[39] RAHIMNEJAD S, YUAN X Y, LIU W B, et al. Evaluation of antioxidant capacity and immunomodulatory effects of yeast hydrolysates for hepatocytes of blunt snout bream (Megalobrama amblycephala) [J]. Fish & Shellfish Immunology, 2020, 106: 142−148.
[40] EL EUONY O I, ELBLEHI S S, ABDEL-LATIF H M, et al. Modulatory role of dietary Thymus vulgaris essential oil and Bacillus subtilis against thiamethoxam-induced hepatorenal damage, oxidative stress, and immunotoxicity in African catfish (Clarias garipenus) [J]. Environmental Science and Pollution Research International, 2020, 27(18): 23108−23128. DOI: 10.1007/s11356-020-08588-5
[41] IGHODARO O M, AKINLOYE O A. First line defence antioxidants-superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPX): Their fundamental role in the entire antioxidant defence grid [J]. Alexandria Journal of Medicine, 2018, 54(4): 287−293. DOI: 10.1016/j.ajme.2017.09.001
[42] 姜会民. 氨氮胁迫对黄河鲤幼鱼肝胰脏、肾脏抗氧化性的影响 [J]. 山东大学学报(理学版), 2012, 47(1):17−22. JIANG H M. Effect of ammonia on atioxidant in the liver, pancreas, and kidney of Yellow River Cyprinus carpio [J]. Journal of Shandong University (Natural Science), 2012, 47(1): 17−22. (in Chinese)
-
期刊类型引用(0)
其他类型引用(4)




 下载:
下载: